AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Atomic number 99/16/2023  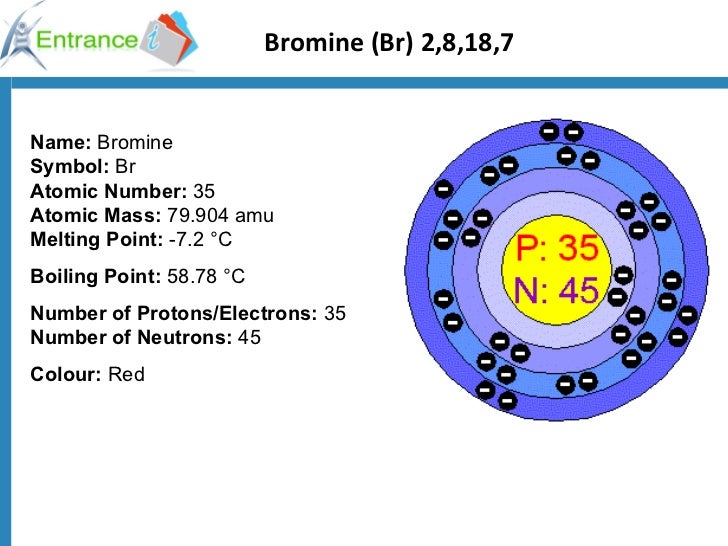
The #p_z# orbital is half-filled because of Pauli's Exclusion Principle, which states that all three #p# orbitals must be half-filled before any of them can be filled completely. For instance, you might have read about it in discussions of nuclear energy, the Fukushima reactor tragedy, or the development of nuclear weapons. Introduction Radioactivity pops up fairly often in the news. This electron will be added to the #2p_z# orbital, which is the #p_z# orbital located on the second energy level.įinally, the spin quantum number, #m_s#, which tells you the spin of the electron, can take two possible valuesīecause, by convention, electrons added to a half-filled orbital are assigned spin-down. The atomic number is the number of protons in an atom, and isotopes have the same atomic number but differ in the number of neutrons. Since the #"9th"# electron corresponds to group #17#, you can say that it will have Atomic Number: 9: Atomic Radius: 135 pm pm (Van der Waals) Atomic Symbol: F: Melting Point:-219.67 ☌: Atomic Weight: 19.00: Boiling Point:-188. #m_l = +1 -># descrbes the #p_x# orbital corresponds to groups #15# and #18#.All of the electrons in the noble gas neon (atomic number 10) are paired, and all of the orbitals in the n 1 and the n 2 shells are filled. #m_l = color(white)(-)0 -># describes the #p_z# orbital corresponds to groups #14# and #17# Fluorine (atomic number 9) has only one 2p orbital containing an unpaired electron.#m_l = -1 -># describes the #p_y# orbital #-># corresponds to groups #13# and #16#.This list contains the 118 elements of chemistry.  
click on any elements name for further chemical properties, environmental data or health effects. Next, for the #p# subshell, the magnetic quantum number, #m_l#, which tells you the orientation of the orbital in which the electron is located, will be equal to Chemical elements listed by atomic number The elements of the periodic table sorted by atomic number. This implies that the angular momentum quantum number, #l#, which describes the energy subshell in which the electron is located, will be equal toĪnd so on. Now, fluorine is located in group #17# of the Periodic Table, which means that fluorine is part of the #p# block. You know that fluorine has an atomic number equal to #9#, which means that the #"9th"# electron in an atom of fluorine is the last one added when "constructing" the neutral atom of fluorine.įluorine is located in period #2# of the Periodic Table, which implies that the #"9th"# electron in fluorine is located on the second energy level.Ĭonsequently, you can say that this electron will have a principal quantum number, #n#, which describes the energy level on which an electron is located in an atom, equal to 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
